
It turns out that thanks to quantum mechanics there's actually a list of possible states of all those things, not just a continuum. That leaves an enormous number of properties of the individual atoms etc. The sort of description used in thermodynamics specifies only a few properties of the system, such as its energy, volume, etc. Those systems (a tube of gas, a block of copper.) have lots of little pieces. Thank you once again.Jack - Jack (age 20) How can you get that from a macroscopic point of view (and particularly from dS = dQ/T)? Sorry for the long question, but I have no idea where I can find those answers except here. 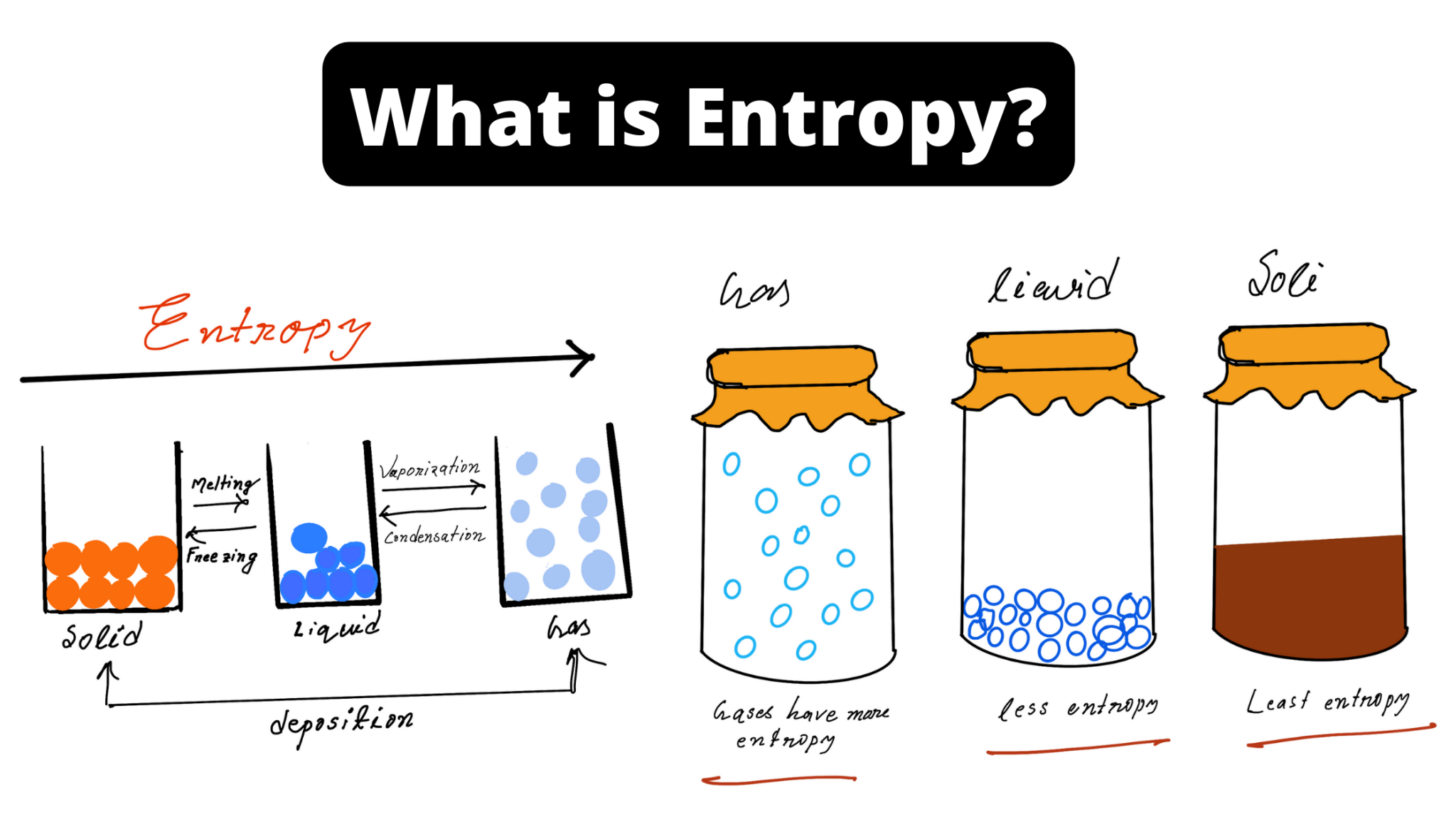
Since there is no energy transfer by heat, does that mean the entropy is equal to zero? In addition, entropy is said to be a measure of disorder or a measure of the quality of the stored energy. However, based on dS = dQ/T, how can you find dQ? Let us assume that we want to know the entropy of an isolated system. I thought I could replace the initial state by one where the entropy is equal to zero, which should give me the entropy of the system. For example, you can calculate the kinetic energy of something directly with 1/2mv�. How do you calculate the entropy of something? I only found examples to calculate a change in entropy. Entropy is a property of a system defined as dS = dQ/T. I have a question regarding entropy from a macroscopic point of view. Hi everyone, First of all, thank you very much for this amazing website.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
